Lab History
 It
has been over a quarter century since Drs. Smerdon and Lieberman published
their groundbreaking paper demonstrating that significant structural
rearrangements occur at the nucleosome level of chromatin during
nucleotide excision repair (NER) of DNA damage in human cells
(Smerdon and
Lieberman, 1978, Proc. Natl. Acad. Sci. USA 75:
4238). Understanding the mechanism(s) of this rearrangement process
has
been one of the goals of the Smerdon lab ever since. It was clear
from the
beginning that DNA repair must take place throughout the genome, within
all levels of the chromatin packaging hierarchy, since no region
within
chromatin is fully protected from exogenous and/or endogenous DNA
damage.
In 1987, the Smerdon lab reported that UV light-Induced DNA damage is
strongly
modulated by the packaging of DNA into nucleosomes (Gale et al., 1987,
Proc.
Natl. Acad. Sci. USA
84: 6644). This report helped set the stage for
our understanding that
both DNA damage and DNA repair can be strongly
modulated
by the packaging of DNA in chromatin.
It
has been over a quarter century since Drs. Smerdon and Lieberman published
their groundbreaking paper demonstrating that significant structural
rearrangements occur at the nucleosome level of chromatin during
nucleotide excision repair (NER) of DNA damage in human cells
(Smerdon and
Lieberman, 1978, Proc. Natl. Acad. Sci. USA 75:
4238). Understanding the mechanism(s) of this rearrangement process
has
been one of the goals of the Smerdon lab ever since. It was clear
from the
beginning that DNA repair must take place throughout the genome, within
all levels of the chromatin packaging hierarchy, since no region
within
chromatin is fully protected from exogenous and/or endogenous DNA
damage.
In 1987, the Smerdon lab reported that UV light-Induced DNA damage is
strongly
modulated by the packaging of DNA into nucleosomes (Gale et al., 1987,
Proc.
Natl. Acad. Sci. USA
84: 6644). This report helped set the stage for
our understanding that
both DNA damage and DNA repair can be strongly
modulated
by the packaging of DNA in chromatin.
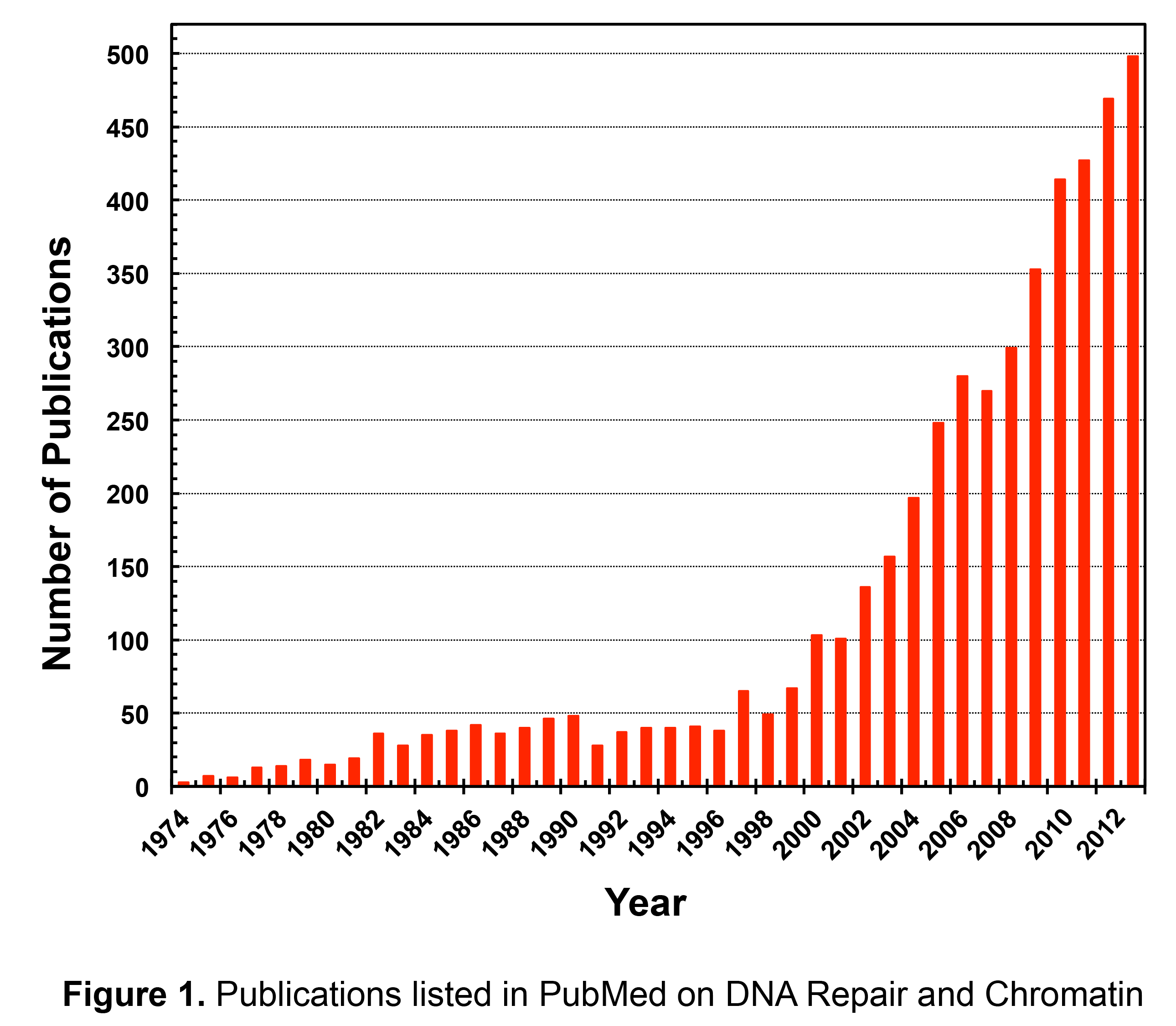
New
technological advances led to a more comprehensive understanding
of these
findings, and during the past decade there was a significant increase in
research focused directly on DNA repair and chromatin (Figure 1).
Some of
this renewed interest was the result of an increased understanding of
how
proteins involved in transcription interact with nucleosomes (e.g., see
Meijer
and Smerdon, 1999, BioEssays
21, 596). There is now convincing evidence that
nucleosome modifications and rearrangements are required for
efficient
transcriptional processing of DNA. Since the late 70s, the Smerdon lab has
focused on correlations between DNA damage, DNA repair, gene transcription
and
chromatin structure. Comprehensive reviews on the work done in this lab
can be
found in Smerdon and Conconi (1999)
Prog.
Nucl. Acid Res. Mol. Biol.,
62:227, and Gong et al. (2005) DNA Repair
4:884.
Smerdon Bio |
Research Statement |
Current Lab Members |
Past Lab Members |
Publications |
Photo Album |
Contact Info |
Home